GB-100 – A Platform Accelerator For Next-Gen Assets
Phase II asset with two orphan indications:
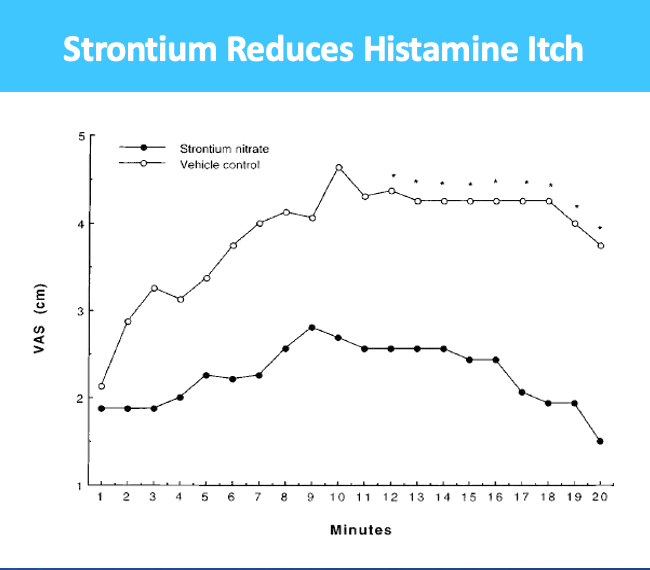
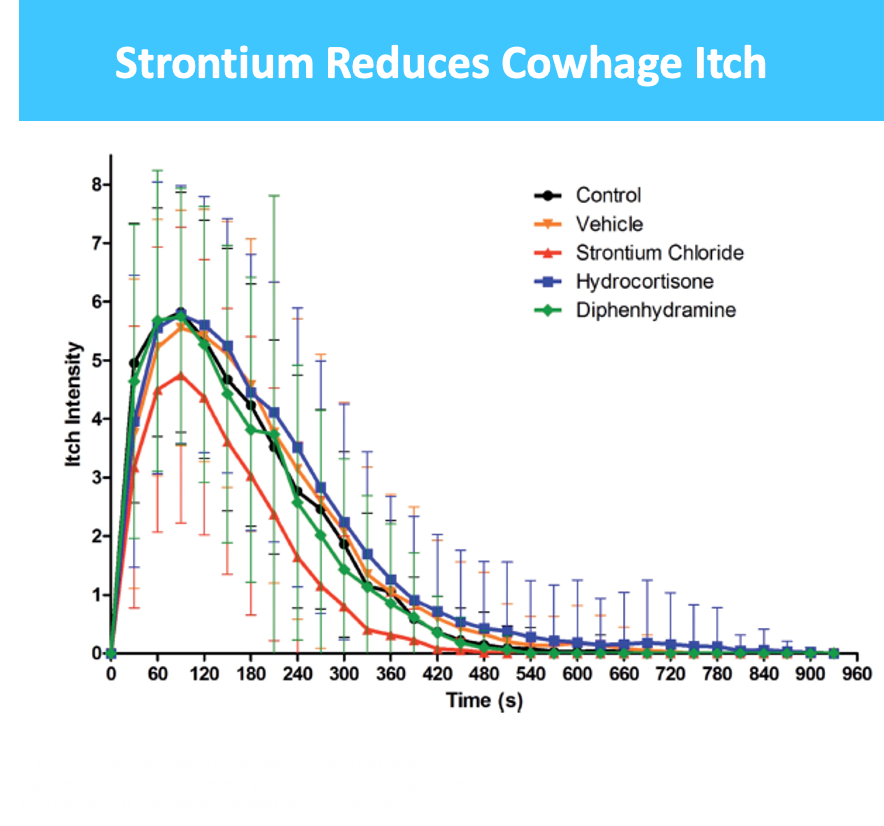
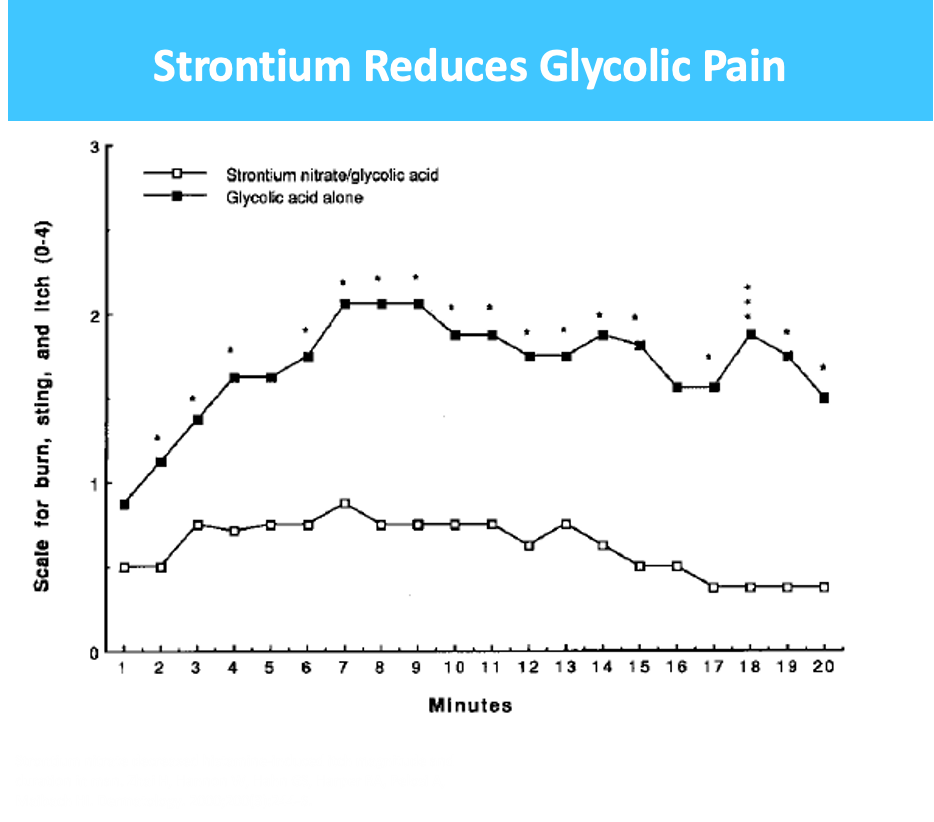
Radiodermatitis (RD) – topical drug for treatment of side effects experienced by 50% of total cancer patients undergoing radiotherapy, 80% to 95% of whom suffer from RD within first week of therapy
- $750M market + 7 and 10 yr orphan exclusivity for US and EU respectively
Chemotherapy-Induced Peripheral Neuropathy (CIPN) – topical drug for treatment of side effects experienced by patients undergoing paclitaxel- or oxaliplatin-based chemotherapy for cancer
- $2.4B market + 7 and 10 yr orphan exclusivity for US and EU respectively
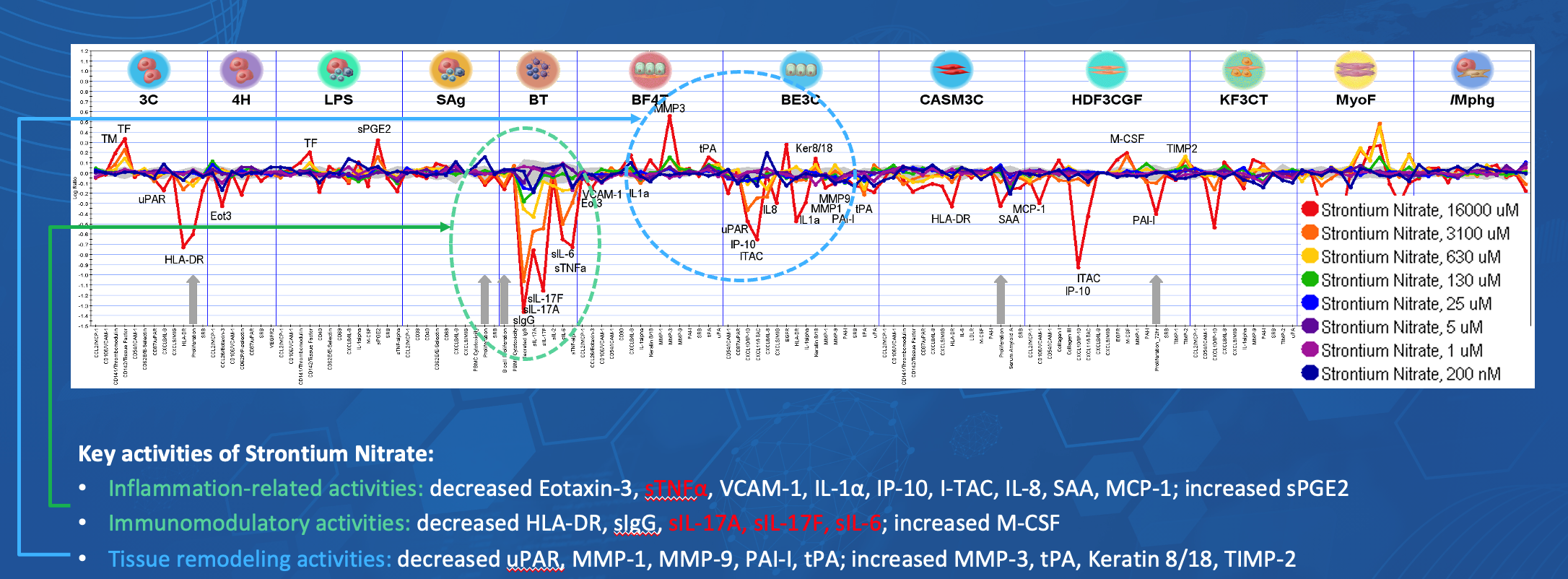
Results from Phase 1a study in healthy volunteers support the safety of GB100 Cream with respect to adverse events, irritation, pain and sensory irritation scores, clinical laboratory tests, and systemic exposure. There were no safety issues associated with the topical application of GB100 at single doses of elemental strontium up to 2 mg/kg
Results from Phase 1b study in patients with Neuropathic pain support that GB100 Cream was safe and well tolerated in subjects with peripheral neuropathic pain with respect to adverse events, irritation, pain and sensory irritation scores, clinical laboratories, and systemic exposure.
Quick Link
Information
Our Location
- 511 S Orange Ave #2069 Newark NJ 07103
Social Media
Copyright © 2024 All rights reserved by GalleonBio
- Privacy Policy